Eculizumab, the monoclonal antibody targeting C5, is the only licensed treatment for Paroxysmal Nocturnal Hemoglobinuria (PNH) in the UK. Inherent to the mechanism of action, C5 inhibitors increase patient susceptibility to encapsulated microorganisms, particularly Neisseria meningitidis. The PNH National service (UK), has 18 years of experience treating patients with PNH using complement inhibition. The risk of N. meningitidis is mitigated by vaccination, ciprofloxacin (500 mg bd) on days 1-13 since we moved to vaccination on day one of complement inhibitor therapy, followed by daily prophylaxis with penicillin (or erythromycin). Since a case of sepsis with penicillin-resistant meningococci was observed, patients also have a rescue course ciprofloxacin. Patient education, safety cards, prompt action in case of fever and a 24 hour on-call service for patients are equally important.
Until 2010 patients were revaccinated with MenACWY every 3 years. Bexsero (MenB vaccine) vaccination (2 vaccines within first 6 months) with boosters every 5 years was added in 2015. In collaboration with the Public Health England Meningococcal Reference Unit in 2010 a program was developed to monitor antibody titers after vaccination and to revaccinate against MenACWY if titers declined to below protective levels. It is technically not possible to assay for meningoccal serogroup B antibody titers when on Eculizumab therapy. We present the outcome of this project.
Methods: Antibody titers to serogroups ACWY were assayed following vaccination and then once per annum. Patients with unprotective antibody titers were revaccinated. We evaluate our practice and review the 9 meningococcal infections in 8 patients. We present disease characteristics, serogroup and outcome, vaccination history and antibody status.
Results: Between May 2002 and July 2020, 324 patients commenced complement inhibitor treatment for PNH. 801 vaccinations with MenACWY were administered; median 2 vaccinations per patient (range 1 - 10). A total of 1,671 antibody titer assessments were conducted in 294 patients, median of 4 tests per patient (range 1 - 15). Every test assessed antibodies against all four serogroups.
Titers were not assessed in 9% of patients (30), due to vaccination prior to change in practice or recent commencement on treatment.
A protective antibody response to all serogroups after first vaccination was observed in 170 / 294 patients (57.8%) and a partial response (antibodies to 3 serotypes) in 51 /294 (17.3%). Revaccination of 51 partial responders resulted in an additional 21 patients with a full response. Revaccination of 73 non-responders (antibodies to 0-2 serotypes) resulted in 32 more partial or full responses.
287 of 324 patients received MenB vaccinations; median 2 vaccinations per patient (range 1 - 4).
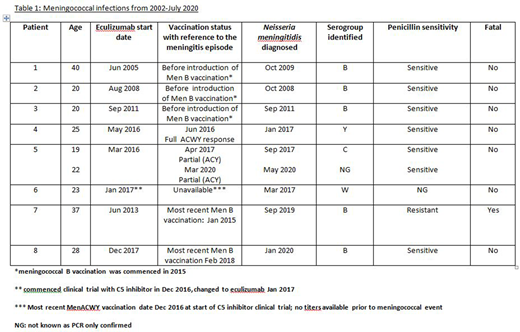
Eight of 324 (2%) patients with median age 22.5 years developed meningococcal sepsis (see table); patient 5 had 2 episodes. 3 of 5 cases with serogroup B infection were before serogroup B vaccination was introduced. The other 4 episodes in 3 patients were due to Y, C, W meningococci, in one the serogroup is unknown. All except patient 1 were compliant with antibiotic prophylaxis. Patient 7 died from meningococcemia, a delay in seeking medical attention may have contributed, however this was also a penicillin resistant strain.
Discussion:
We report the largest experience of managing meningococcal risk in patients on complement inhibitor therapy for PNH. Despite our proactive management we had 9 cases of meningococcal sepsis, with one fatal infection. Our most recently introduced practice of prompt treatment with ciprofloxacin if pyrexic on antibiotic prophylaxis will prevent cases like patient 7 with a penicillin resistant strain. Three patients had a meningococci sepsis with serogroups C, W and Y; whilst 1 patient had no check of titers due to recent commencement on treatment, the titres of the other 2 had suggested protective immunity.
We demonstrated that a full antibody response can be obtained on a second vaccination in most patients if the first one failed. If no response is achieved upfront or revaccination then further MenACWY vaccination is not likely to be successful. Current practice significantly mitigates the risk of meningococcal disease, however it is essential patients remain vigilant for fever, seeking immediate medical attention stating their diagnosis of PNH on complement inhibitor therapy.
Arnold:Alexion Pharmaceuticals: Honoraria. Borrow:Pfizer: Research Funding; GlaxoSmithKline: Research Funding; Alexion pharmacueticals: Research Funding; Sanofi: Research Funding. Riley:Alexion: Honoraria. Munir:Alexion: Honoraria; F. Hoffmann-La Roche: Consultancy, Other: Medical writing support, furnished by Scott Battle, PhD, of Health Interactions, was funded by F. Hoffmann-La Roche Ltd, Basel, Switzerland. Kelly:Alexion: Honoraria. Pike:Apellis: Research Funding. Hillmen:Acerta: Other: Financial or material support; Roche: Consultancy, Other: Financial or material support, Research Funding, Speakers Bureau; AbbVie: Consultancy, Other: Financial or material support, Research Funding, Speakers Bureau; Pharmacyclics: Other: Financial or material support, Research Funding; Janssen: Consultancy, Other: Financial or material support, Research Funding, Speakers Bureau; AstraZeneca: Consultancy, Speakers Bureau; Gilead: Other: Financial or material support, Research Funding; Alexion: Consultancy, Research Funding, Speakers Bureau; Apellis: Consultancy, Research Funding, Speakers Bureau. Griffin:Alexion Pharmaceuticals: Honoraria, Other: Conference Support; Biocryst: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal